
As the nation comes to terms with the possibility of widespread COVID-19 infections, announcements about new guidelines and regulation changes are being made daily. The first COVID-19 relief bill (H.R. 6074), signed by President Trump on March 6 and summarized here by ECG, expanded access to telehealth services for Medicare beneficiaries, reduced restrictions related to the originating site of care, and provided the ability to receive a telehealth screening via smartphone. On March 17, the president and CMS announced additional clarification and guidance for H.R. 6074 that will be critical for providers interested in using telehealth to screen for and treat COVID-19.
Expansion of Telehealth with 1135 Waiver
One noteworthy limitation of the March 6 legislation was that in order for a Medicare beneficiary to receive telehealth services, they were required to have an established relationship with the provider. The patient also needed to have received a Medicare-covered service within the last three years from the provider or another provider in the same practice. CMS has now announced that audits will not be conducted to ensure a prior relationship existed, thus removing a potential barrier to receiving care.
Under the new CMS guidelines and as a tactic to reduce the risk of exposure to COVID-19, Medicare beneficiaries can now also receive an expanded list of services via telehealth, including E&M visits, behavioral health, and preventive health screenings. All telehealth visits will be reimbursed at the same rate as in-person visits.
While these new telehealth benefits apply to traditional Medicare, Medicare Advantage plans continue to follow guidelines published by CMS last week. These guidelines provide increased flexibilities in regard to waiving cost sharing of COVID-19 treatment, including telehealth, and expanding telehealth access. While opportunities exist to offer additional telehealth services, payers continue to have discretion in their service offerings. Therefore, providers should work closely with Medicare Advantage plans to clarify and confirm that they will follow these new CMS guidelines.
Types of Virtual Services
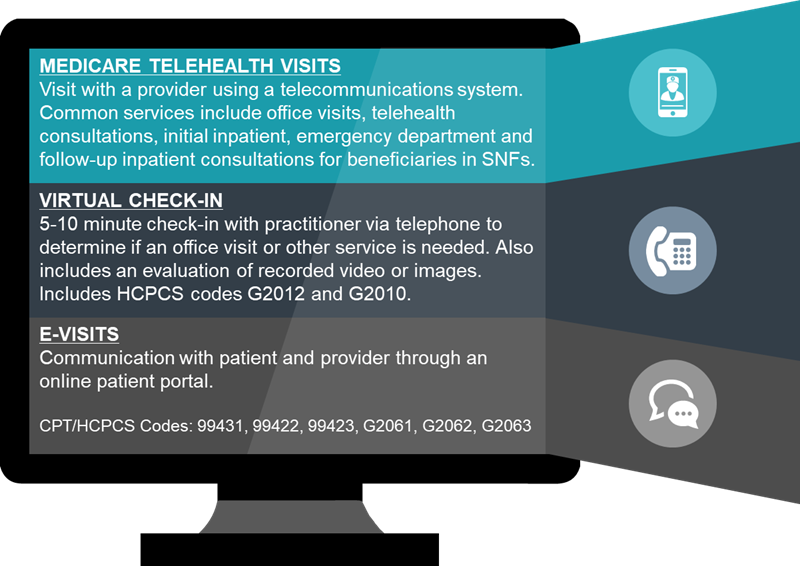
The March 17 CMS guidance identified and differentiated three types of COVID-19 virtual services that clinicians can provide to Medicare beneficiaries under the new 1135 waiver, regardless of beneficiary geography.
Telehealth Visits: Providers can use an interactive audio/video telecommunications system that permits real-time patient communication for office visits, hospital visits, and other services that would otherwise generally occur as an in-person encounter. Importantly, while CMS notes the previous requirement that Medicare beneficiaries have a prior established relationship with eligible providers, CMS explicitly states that HHS will not conduct any audits to ensure that the required prior relationship existed for any claims submitted during this public health emergency. CMS provides the full list of 101 codes that can be provided via telehealth online for reference.
Virtual Check-Ins: A brief patient interaction with a clinician can occur via a number of communication modalities, including synchronous discussion over a telephone or exchange of information through video or image. This method of service is meant for patients with an established relationship with the provider, and where the communication is not related to a medical visit within the previous seven days and does not lead to a medical visit within the next 24 hours. Existing Medicare coinsurance and deductibles would apply, and patients must verbally consent to receive a virtual check-in.
E-Visits: Non–face-to-face patient-initiated communications or online evaluation and management with an eligible clinician can occur via the use of an online patient portal over a seven-day period. Similar to virtual check-ins, patients must have a previously established relationship with the provider and have initiated the virtual inquiry.
For more information and additional specifics of these telehealth rule changes, please see the CMS fact sheet.
HIPAA Violation Exceptions
In an unprecedented move that highlights the gravity of the COVID-19 situation, HHS has agreed to “exercise discretion and waive penalties” for any HIPAA violations committed by healthcare providers who “serve patients in good faith through everyday communication technologies such as FaceTime or Skype” during the recognized national emergency time period.
Looking Forward
While these new federal guidelines can greatly expand providers’ ability to appropriately screen and treat patients through telehealth, it is necessary to note that state-specific regulations are still active and complexities will continue to be identified.
Additionally, H.R. 6074 was the first of at least three large emergency COVID-19 legislative actions meant to help stimulate the American economy and provide support for COVID-19 screening and treatment. At least two more bills are also expected to pass through Congress for President Trump to sign in the coming days that will likely have additional implications for telehealth screening and treatment, including potential reimbursement guidance for commercially insured or uninsured patients. We expect to see continued updates related to telehealth services as a result of changes occurring on a state level, additional CMS guidelines, and new federal legislation.

